Article 11 in the Got Real Milk Series
In April 2026, the U.S. Food and Drug Administration (FDA), under the Department of Health and Human Services (HHS), released the results of a market-basket analysis of infant formula products in the United States. A total of 312 samples from 16 brands were evaluated for a range of environmental contaminants, including heavy metals, per- and polyfluoroalkyl substances (PFAS), pesticides, and plasticizers.
The agency concluded that contaminant levels were “low” and generally within established safety thresholds. While this conclusion is factually grounded within the framework of regulatory toxicology, it warrants closer examination particularly when interpreted through a pediatric and exposure-based lens.
This analysis does not challenge the validity of the laboratory methods employed. Rather, it highlights the gap between analytical detection and clinical interpretation, especially in the context of infant health.
Scope of Testing and Principal Findings
The FDA evaluated infant formula “as prepared for feeding,” an important methodological choice that reflects real-world consumption patterns.
Analytes included:
- Heavy metals: arsenic, lead, cadmium, mercury
- PFAS (30 targeted compounds)
- Pesticides (including glyphosate and glufosinate)
- Phthalates and related plasticizers
Key findings included:
- Lead detected in approximately 80% of samples (maximum 1.1 ppb)
- Arsenic detected in approximately 94% (maximum 4.7 ppb; subset identified as inorganic arsenic)
- Cadmium detected in approximately 66% (maximum 1.5 ppb)
- PFOS detected in roughly 50% of samples
- Phthalates detected in approximately 46%
Mercury and most pesticides were largely not detected.
At a population level, these findings may be interpreted as reassuring. However, such interpretation is contingent upon how risk is defined and for infants, this requires a distinct framework.
Limitations of Data Presentation
The FDA presented results using categorical concentration ranges (e.g., 0.3–0.5 ppb; 0.6–2.0 ppb). While visually accessible, this approach introduces several interpretive limitations:
- Absence of distribution within categories
- Lack of product-specific identification
- No indication of clustering toward upper or lower bounds
- Inability to assess variability across brands or batches
From a toxicological standpoint, the difference between values at the lower versus upper end of a range may be clinically meaningful. Without this differentiation interpretation is limited.
Exposure Assessment: The Missing Dimension
Perhaps the most significant limitation of the FDA analysis is the absence of dose-based exposure modeling.
Infants do not ingest contaminants as concentrations alone; they ingest volume. A typical formula-fed infant consumes approximately:
- 24–32 ounces per day
- Equivalent to 7–0.95 liters daily
When translated into daily intake, even low concentrations result in repeated, cumulative exposure during a vulnerable developmental period.
Despite this, the FDA does not present results in terms of:
- Micrograms per day (µg/day)
- Micrograms per kilogram body weight (µg/kg/day)
These metrics are fundamental to pediatric risk assessment.
Toxicological Considerations in Early Life
Infants represent a uniquely vulnerable population due to:
- Rapid neurological and organ development
- Immature renal and detoxification systems
- Reliance on a single primary food source
Several contaminants identified in this analysis are known health toxicants. Note the following:
- Lead: No known safe level for neurodevelopment
- Inorganic arsenic: Neurotoxic and carcinogenic
- Cadmium: Associated with renal and skeletal effects
- PFAS: Persistent, bioaccumulative, and immunologically active
- Phthalates: Endocrine-disrupting compounds
Regulatory comparisons are frequently made against drinking water standards or adult-based thresholds. However, such comparisons may not adequately reflect infant-specific susceptibility or exposure patterns.
Unaddressed Questions
Despite the breadth of testing, several key questions remain:
- Which specific products contained higher contaminant levels?
- Do multiple contaminants exert additive or synergistic effects?
- What is the cumulative exposure burden over time?
- How do measured exposures compare to infant-specific safety benchmarks?
- Why are adult-based reference standards applied to infant populations?
These questions are central to translating analytical findings into meaningful health guidance.
Notable Omissions in Testing Scope
Two additional limitations merit attention:
Lack of Differentiation Between Organic vs. Conventional Formulas
The analysis does not distinguish between organic and conventional infant formula. Given known differences in agricultural practices, pesticide exposures, and contamination pathways, this omission limits parents’ ability to navigate the supermarket shelves when deciding what is best for their infants.
Absence of Aluminum Testing
Aluminum, a known neurotoxicant documented to be contaminated in infant formula (particularly soy-based and specialized formulas), was not included in the testing panel. This is concerning given:
- Established literature on aluminum accumulation in infants
- Associations with neurodevelopmental and bone-related effects
- Increased vulnerability due to immature renal function
The exclusion of aluminum represents a significant gap in comprehensive contaminant assessment.
In our infant formula study published May, 2024, , we found aluminum levels as high as 41,000 ppb in one goat milk formula. The following chart summarizes our findings which were more concerning as compared to the FDA study:
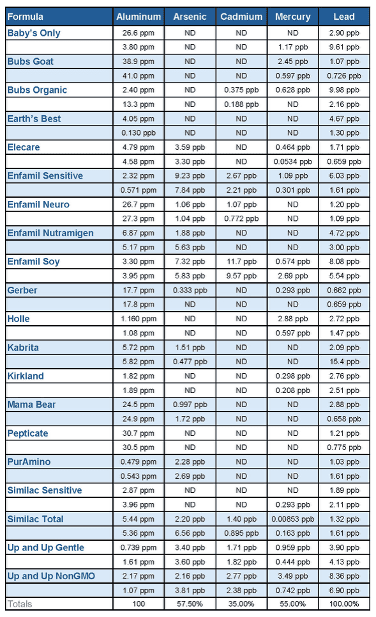
Why the Difference Between the Studies?
One reasonable interpretation of this difference is that increased scrutiny has driven improvements in manufacturing practices.
Over the past several years:
- Independent investigations (including those by GMOScience and Consumer Reports) have brought widespread attention to contaminants in infant formula
- Public and regulatory pressure has increased
- The FDA launched expanded oversight initiatives and large-scale testing efforts
Manufacturers may have been incentivized, whether through regulatory pressure, public visibility, or market forces, to reduce contaminant levels in their products.
Why This Matters
If contaminant levels have indeed declined over time, this carries two important implications:
- Improvement is possible
The presence of lower levels in more recent testing indicates that contamination is not inevitable, and it can be mitigated through sourcing, processing, and quality control. - Past exposure still matters
Millions of infants may have been exposed to higher contaminant levels prior to these improvements, highlighting the importance of ongoing monitoring and transparency.
The contrast between earlier independent testing and more recent FDA findings does not invalidate either dataset. Instead, it highlights a reality:
Infant formula safety is dynamic, not static, and continuous oversight is essential.
Reframing the Findings
The FDA’s conclusions are not inherently incorrect, but they are incomplete when applied to individual-level pediatric risk.
A more precise and crucial interpretation:
At a population level, contaminant concentrations in infant formula are generally low. At the individual infant level, cumulative exposure and developmental impact remain insufficiently characterized.
Future Directions
Advancing infant health protection will require a shift from concentration-based reporting to exposure-informed science, including:
- Infant-specific dose modeling (µg/kg/day)
- Batch-level transparency and public reporting
- Identification of higher-exposure products
- Assessment of cumulative and mixture toxicity
- Regulatory thresholds grounded in developmental biology
Such measures would better align analytics and clinical relevance. In parallel, the State of California is currently evaluating legislation I authored and introduced by Assemblymember Rodriguez that would require infant formula manufacturers to test for and disclose levels of toxic metals. This represents an important first step toward transparency. However, progress will require federal involvement. The Department of Health and Human Services must adopt comparable standards to ensure nationwide consistent accountability. Ultimately, accountability must extend beyond acknowledgment and create enforceable action.
Our babies can’t wait.
Conclusion
Infant formula remains an essential and often the only nutritional source. However, when evaluating environmental contaminants:
“Low levels” do not equate to “no biological impact,” particularly under conditions of daily, cumulative exposure during early development.
Bridging the gap between detection and interpretation is essential to ensuring that regulatory reassurances reflect the realities of infant physiology and development.
References
- 1, U.S. Food and Drug Administration (FDA). Infant Formula Product Testing Results. April 2026. https://www.fda.gov/media/192087/download
- Bishop NJ, Morley R, Day JP, Lucas A. Aluminum neurotoxicity in preterm infants receiving intravenous-feeding solutions. N Engl J Med. 1997;336:1557–1561.
- https://gmoscience.org/2024/05/20/why-study-toxic-metals-in-infant-formula-an-overview-of-the-results/



